How to react to Deficiencies after an FDA Inspection?
Recommendation
Process Validation - Live Online Training
GMP deficiencies are sometimes found during FDA inspections. It is important to respond to this deficiency report, otherwise a Warning Letter may be issued. But how should one respond?
The example of a manufacturer who had no validation of his manufacturing process is a good illustration of how not to respond to FDA "findings". Besides the lack of process validation, the FDA also criticised the lack of qualification protocols and reports as well as the lack of acceptance criteria showing that the manufacturing process was in "state of control". Furthermore, the holding time of the bulk was not validated.
As a remedy, the company said it would hire a third party to assist with validation. This was not enough for the FDA.
The authority demanded:
- A detailed overview of the validation programme with the appropriate written instructions
- A detailed programme for the development, validation, maintenance, control and monitoring of each manufacturing process
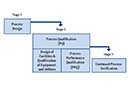
- The process performance qualification (PPQ) programme
- The continuing monitoring programme for intra-batch and inter-batch variability
- A timeline for the implementation of PPQ for each marketed product
- PPQ protocol(s)
- A programme for equipment and facility qualification and written instructions for these activities
For bulk holding times, the FDA requested a description of how time limits are set for these. In addition, a retrospective evaluation of manufacturing activities is required with the aim of identifying possible effects on the stability and quality of the medicinal products.
Conclusion: In response to "findings", the FDA wants to see detailed documents on how these "findings" are remedied. Furthermore, it is often necessary to look back to see whether the deficiencies have an impact on the products manufactured to date.
Please see the Warning Letter issued to System Kosmetik Produktionsgesellschaft für kosmetische Gmbh on the FDA website.